Transforming Respiratory Clinical Trial Recruitment
At Trialbee, we connect people living with chronic and acute respiratory conditions to innovative clinical trials designed to improve breathing, function, and overall quality of life. Through patient-centric strategies and data-driven insights, we help sponsors accelerate recruitment, broaden representation, and deliver new respiratory therapies to patients faster.

Key Challenges in Respiratory Trials
Respiratory studies face significant recruitment challenges — from diagnostic uncertainty and fluctuating symptoms to comorbidities that complicate eligibility. These barriers slow enrollment, reduce diversity, and increase study costs.

The result: slower site activation, inconsistent referral-to-randomization rates, and missed opportunities to engage well-matched participants who could benefit from research involvement.
Trialbee’s Solution
At Trialbee, we deliver patient-focused, technology-driven recruitment strategies tailored specifically for respiratory clinical trials. Our digital ecosystem combines omnichannel outreach, medical pre-qualification, and transparent analytics to accelerate enrollment without compromising quality.
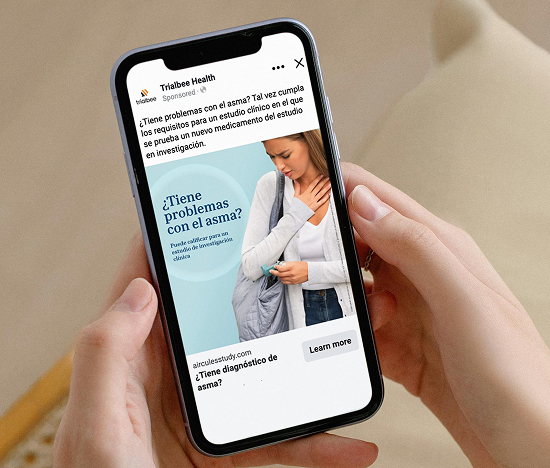
Precision Recruitment & Omnichannel Reach
We connect with respiratory patients where they seek information and support — online communities, advocacy groups, social networks, and health-related platforms. Through behavioral insights, real-world data, and targeted digital engagement, Trialbee identifies and reaches verified, motivated individuals likely to meet study requirements.

Two-Stage Medical Pre-Qualification
All referred patients complete digital pre-screening and structured eligibility verification based on:
- Symptom burden and exacerbation history
- Current and past inhaled/biologic therapies
- Diagnostic details (spirometry, clinician diagnosis, biomarkers where relevant)
- Known triggers, smoking history, and comorbidities
This ensures sites receive pre-qualified, well-informed participants who understand the study and are prepared to engage.

Real-Time Visibility & Analytics with Honey™ Platform
Our Honey™ platform provides clear, real-time insights into enrollment performance and patient progression. Sponsors and CROs can:
- Track recruitment funnel metrics at every stage
- Identify bottlenecks across channels and sites
- Forecast performance and make data-driven adjustments
- Maintain oversight of participant quality and site readiness
Proven Experience Across Key Indications
- Alpha1 antitrypsin deficiency (AATD)
- Chronic Thromboembolic Pulmonary Hypertension (CTEPH)
- Nasal Polyps
- Asthma
- Cough Mint
- Progressive Pulmonary Fibrosis
- Asthma (Adolescent)
- Idiopathic Pulmonary Fibrosis
- Sleep Apnea
- Chronic obstructive pulmonary disease (COPD)
- Sleep Apnea + Obesity/Overweight



Creative that Truly Resonates
Respiratory conditions impact daily life — from physical activity and sleep to work, social interactions, and emotional well-being. Trialbee develops empathetic, inclusive campaigns that:
- Reflect the real lived experiences of respiratory patients
- Communicate complex conditions simply and clearly
- Emphasize confidence, empowerment, and improved quality of life
Our creative approach centers authenticity and accessibility, ensuring every message resonates with the respiratory community and encourages participation with dignity and trust.
Let’s Partner for Better, Faster Recruitment
Whether you’re conducting a study in inflammatory bowel disease, liver conditions, or rare gastrointestinal disorders, Trialbee helps you connect with qualified patients — faster. Learn how our technology, data, and human-centered approach deliver measurable recruitment success.


